Finding the Molar Mass of an Element:
To find the molar mass of an element, you'd have to look it up on the periodic table. In the periodic table below, the molar masses are displayed below the symbol of each element:
 |
| click to enlarge |
Calculating the Molar Mass of a Molecule
To calculate the molar mass of a molecule, you have to first find the molar mass of each individual element. Then, you have to multiply each molar mass by its element's subscript. After, add the values together and the molar mass of the molecule will be obtained. For example, to calculate the molar mass of water (H2O):
- Find molar mass of each element:
- Oxygen : 15.999
- Hydrogen :1.0079
- Multiply each molar mass by the subscript of the element (H2O1):
- Oxygen: 15.999×1= 15.999
- Hydrogen: 1.0079×2= 2.0158
- Add the results together:
- 15.999+2.0158= 18.0148 grams/mole
Converting Molar Mass to Mass:
To find the mass of a certain amount of molecules, use the following equations
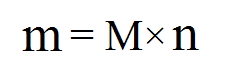
- m: mass of the molecule
- M: molar mass of the molecule
- n: the number of moles
This is quite simple. Imagine that you have three bags of tomatoes and each of them weigh 2 kilograms. To calculate the total mass, you'd multiply the mass per bag (2 kg) by the number of bags (3 bags), and you would get 6 kilograms as your answer.
Similarly, if you have three moles of a molecule, and you know that the molar mass of the molecule is 2 grams/mole, then you would multiply 2 by 3 to get 6 grams as the total mass.
You can also change the equation a bit to calculate the number of moles or the molar mass (if you have the other two variables):
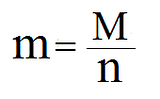
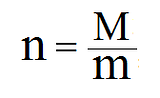
Worksheets:
The concept of molar mass is extremely important in stoichiometry, and make sure that you know how to do the problems in these two worksheets:
- Finding molar mass: http://misterguch.brinkster.net/PRA001.pdf
- Using molar mass in equations: http://misterguch.brinkster.net/PRA050.pdf
