For the next few weeks or so, I will be extremely busy— busier than I ever was. I will have to complete four to six tasks each day, and each of them will take at least half an hour (and as much as 2-3 hours). So starting tomorrow, I must not even waste a minute on television, Internet-surfing, or games. I also have to make sure that I am constantly thinking about my projects so that they will not have poor qualities. Otherwise, my marks will suffer and my average will be even lower than it is now.
Rant: My English teacher is driving me crazy! Five *big* assignments in 20 days!? How's that giving us "ample amount of time" for each assignment?
Thursday, November 24, 2011
Monday, November 21, 2011
Chemical Equilibrium #4: Manipulating the Equilibrium Constant (keq)
When calculating the keq of the reverse reaction:
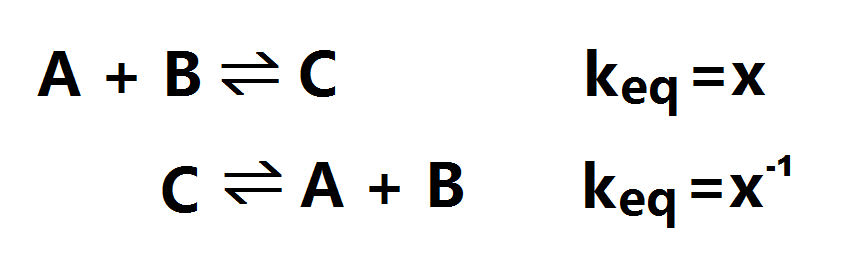
When multiplying the entire equation by a number:
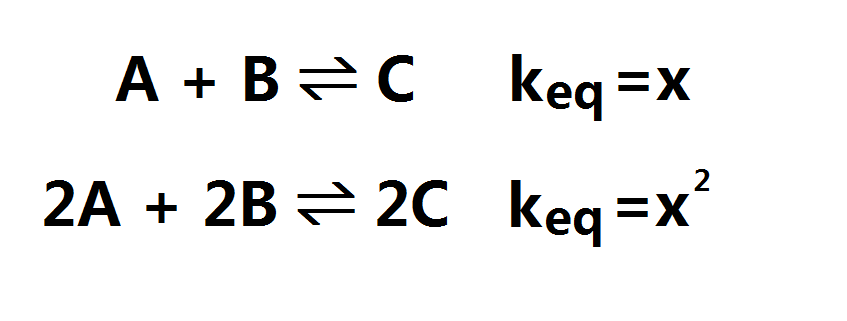
When adding several equations together:
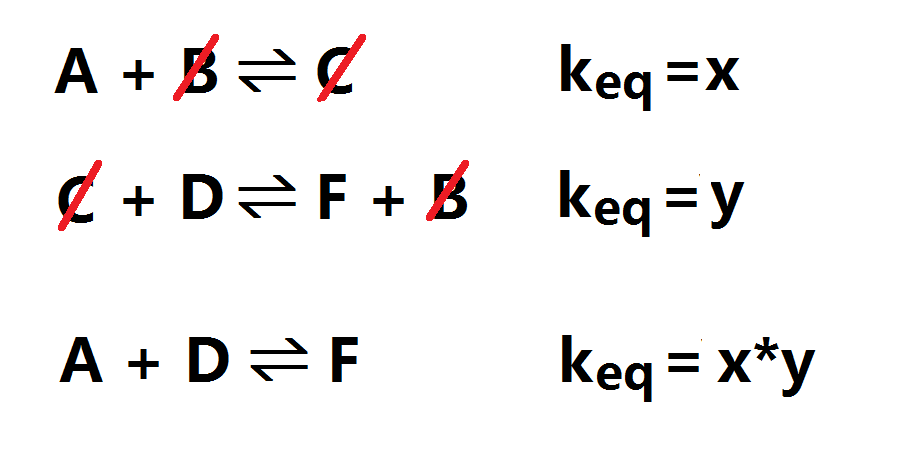
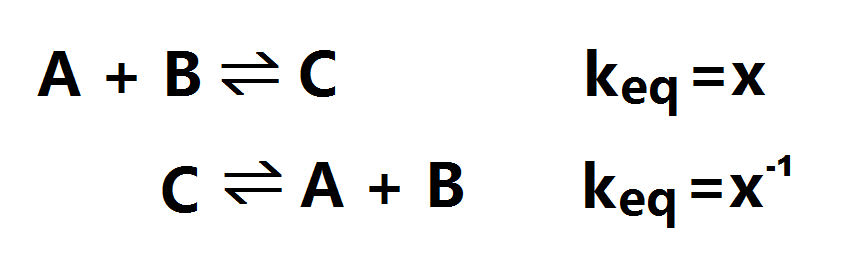
When multiplying the entire equation by a number:
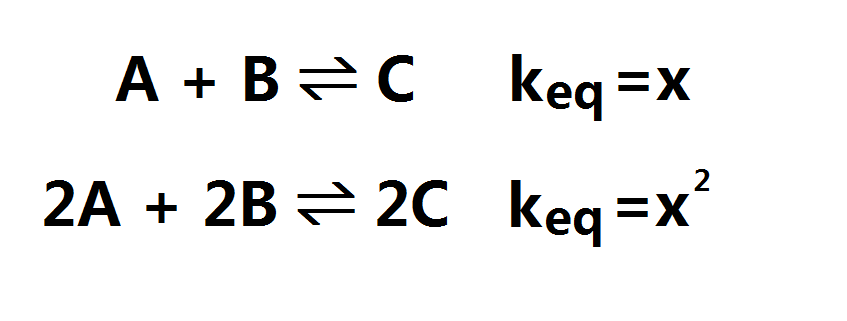
When adding several equations together:
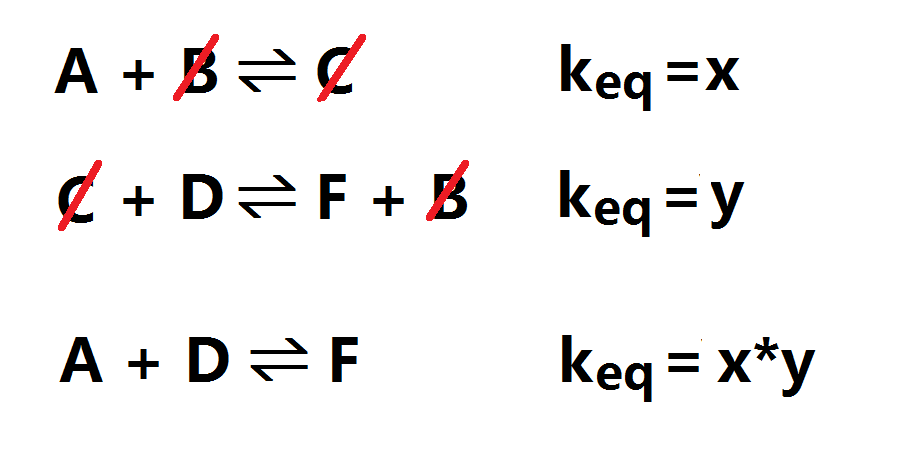
Labels:
Chemistry
Chemical Equilibrium #3: Equilibrium Constants
In a closed chemical system at constant temperature, there's a constant (keq) that defines the relationships between the concentrations of products and reactants.
Keq= (concentration of each product raised to a power of the coefficient of the corresponding product)÷ (concentration of each reactant raised to the coefficient of the corresponding reactant).
To make it more clear,

Keq= (concentration of each product raised to a power of the coefficient of the corresponding product)÷ (concentration of each reactant raised to the coefficient of the corresponding reactant).
To make it more clear,

(where the lower case letters are the coefficients)
What keq tells:
Since keq is products over reactants, the larger it is, we know that the more products there are in the equilibrium (and vice versa). So a large keq (e.g., 1000000) means that the reaction is close to completion, and a very small keq (e.g., 0.00000001) means that the reaction barely occurs.
Since keq is products over reactants, the larger it is, we know that the more products there are in the equilibrium (and vice versa). So a large keq (e.g., 1000000) means that the reaction is close to completion, and a very small keq (e.g., 0.00000001) means that the reaction barely occurs.
Labels:
Chemistry
Chemical Equilibrium #2: Le Chatelier's Principle
Le Chatelier's Principle predicts what would happen to a system at equilibrium if certain conditions are changed. The general statement is:
Change in Concentration
In the above reaction, if the concentration of C were increased, then the equilibrium must shift to counteract this change. In order to do so, the equilibrium will have to shift toward the reverse direction so that the excess C can be consumed.
If, instead, the concentration of C were decreased, then the opposite will happen. The reaction will have to shift toward the forward direction so that there can be more C.
If a chemical system at equilibrium experiences a change in concentration, temperature, pressure or volume, the equilibrium will be shifted to counteract this change, and a new equilibrium will be established.Now, let's break this statement down:
Change in Concentration
A + B ⇌ C + D
In the above reaction, if the concentration of C were increased, then the equilibrium must shift to counteract this change. In order to do so, the equilibrium will have to shift toward the reverse direction so that the excess C can be consumed.
If, instead, the concentration of C were decreased, then the opposite will happen. The reaction will have to shift toward the forward direction so that there can be more C.
Labels:
Chemistry
Chemical Equilibrium #1: Introduction
What is equilibrium?
Equilibrium is the state of a closed system at which two opposing reactions are simultaneously occurring at the same rate. In other words, in an equilibrium, the concentrations of all chemicals are constant.
(Note: even though the two reaction rates are equal to each other, it doesn't mean that the concentrations of the chemicals are equal)
Equilibrium is the state of a closed system at which two opposing reactions are simultaneously occurring at the same rate. In other words, in an equilibrium, the concentrations of all chemicals are constant.
(Note: even though the two reaction rates are equal to each other, it doesn't mean that the concentrations of the chemicals are equal)
Labels:
Chemistry
Saturday, November 19, 2011
Chemistry: How to Solve Titration Questions
Strong Acid + Strong Base Titration
- Convert concentrations to number of moles
- Subtract the smaller n from the larger n
- Calculate -log (difference÷total volume)
- If the larger n is of the acid, the result is the pH. Otherwise, it's the pOH.
Weak Acid+ Strong Base Tritation
Labels:
Chemistry
Thursday, November 17, 2011
Dream: What is Fibonacci?

Labels:
dreams
Novel for English Class
Today it was revealed that we'd be reading The Picture of Dorian Gray by Oscar Wilde as the novel for English class. Strange, because I am pretty sure that at the beginning of September, the teacher said that we'd be reading The Kite Runner by Khaled Hosseini.
But this is actually good, because I've already read The Kite Runner two years ago, and it would be sort of boring if I were to read it twice. Also, as The Picture of Dorian Gray has quite a high rating from various websites, I am looking forward to reading it.
So yeah, I hope I will be able to enjoy this book.
But this is actually good, because I've already read The Kite Runner two years ago, and it would be sort of boring if I were to read it twice. Also, as The Picture of Dorian Gray has quite a high rating from various websites, I am looking forward to reading it.
So yeah, I hope I will be able to enjoy this book.
Labels:
high school
Subscribe to:
Posts (Atom)
